Meeting Documents
The Ocean’s Hidden Buffer: Deep-Ocean Benthic Alkalinity Fluxes, CO2 Neutralisation Potential, and an Emerging Anthropogenic Fingerprint
Presented at: Ocean Sciences Meeting 2026
Abstract
The ocean’s gigatonne-scale CO2 uptake depends not only on physics and biology at the surface, but also on benthic processes that supply total alkalinity (TA) through exchange at the sediment–water interface, thereby modulating the carbonate buffer. Yet the magnitude, spatial structure, and anthropogenic change of benthic TA fluxes remain poorly constrained and rely on empirical upscaling. Here, we generate a global time series (1992–2019) of deep-ocean (>200 m) benthic TA flux by coupling a field-tuned diagenetic sediment model (RADIv2) to the ECCO–Darwin state estimate, and validate it against sediment-trap syntheses, gridded bottom-water carbonate chemistry and in situ flux observations. We then map global benthic TA fluxes, estimate their CO2-neutralisation potential, and separate anthropogenic signals from natural variability in carbonate dynamics and seafloor TA sources. We find that deep-ocean sediments supply a TA efflux whose neutralisation potential is about one-half of today’s net ocean CO2 sink; however, its near-term expression is limited by deep-ocean ventilation timescales.
To isolate anthropogenic change, we run two experiments: (i) a historical simulation with evolving atmospheric CO2 and physical–biogeochemical forcings, and (ii) a no-trend control that repeats a detrended annual cycle of atmospheric CO2, preserving seasonality but holding long-term means fixed. Relative to the control, carbonate kinetics shift: the calcite saturation horizon shoals and bottom-water undersaturation becomes more frequent, increasing CaCO3 dissolution and boosting benthic TA input, while enhanced water-column dissolution reduces carbonate delivery to the seafloor. These results reveal a global anthropogenic fingerprint on the ocean carbonate system and benthic buffering, with implications for the ocean’s long-term buffer capacity.
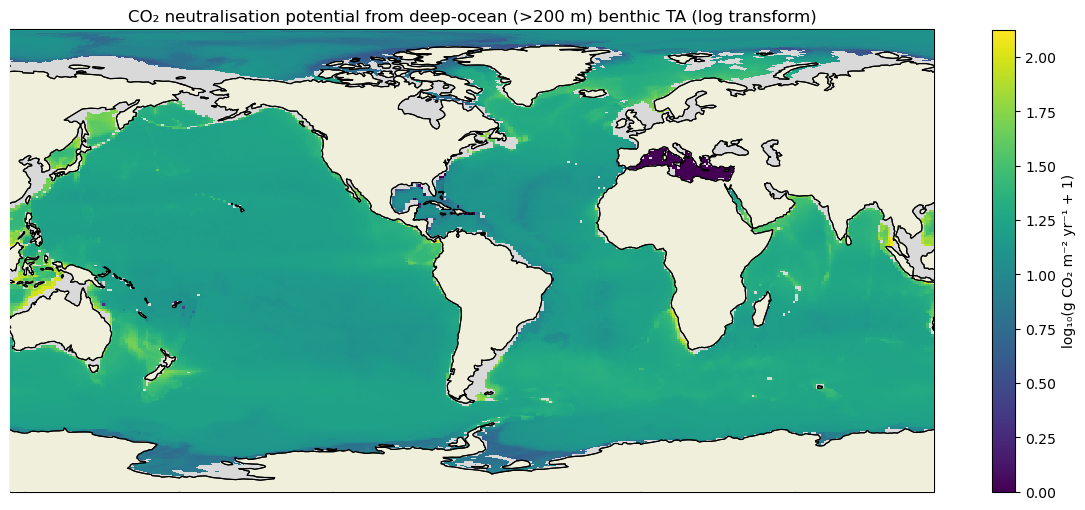
Plain-language Summary: The ocean helps slow climate change by taking up carbon dioxide (CO2) from the air. Part of this ability comes from the seafloor: chemical reactions in sediments release alkalinity, a natural buffer that lets seawater hold more CO2 without large changes in acidity. We built a global picture (1992–2019) of this seafloor alkalinity supply using models that were checked against real-world measurements. We also estimated how much of the ocean’s CO2 uptake this added alkalinity could, in principle, counteract. To see the human influence, we compared two simulations: one with the actual rise in atmospheric CO2 and one with long-term trends removed. We find a clear human-driven signal: as the ocean absorbs more CO2, deep waters become more corrosive, causing shell-forming minerals to dissolve faster and release more alkalinity. This deep-ocean buffering builds over many decades to centuries, but is important for future climate projections.
View Document (OSM26) »
